 |
| The researchers demonstrated how the methods used to create their membranes allow for fine-tuning the spacing of the nanostructures within the resulting filter. Credit: University of Pennsylvania |
Chemical separation processes are essential in the manufacturing of many products from gasoline to whiskey. Such processes are energetically costly, accounting for approximately 10–15 percent of global energy consumption. In particular, the use of so-called "thermal separation processes," such as distillation for separating petroleum-based hydrocarbons, is deeply ingrained in the chemical industry and has a very large associated energy footprint. Membrane-based separation processes have the potential to reduce such energy consumption significantly.
Membrane filtration processes that separate contaminants from the air we breathe and the water we drink have become commonplace. However, membrane technologies for separating hydrocarbon and other organic materials are far less developed.
Penn Engineers are developing new membranes for energy-efficient organic separations by rethinking their physical structure on the nanoscale.
Nanofiltration using self-assembling membranes has been a major research area for Chinedum Osuji, Eduardo D. Glandt Presidential Professor in the Department of Chemical and Biomolecular Engineering, and his lab. The performance of these membranes was highlighted in a previous study describing how the structure of the membrane itself helped to minimize the limiting tradeoff between selectivity and permeability that is encountered in traditional nanofiltration membranes. This technology was also included in last year's Y-Prize competition, and the winners have advanced a case for its use to produce non-alcoholic beer and wine in a startup called LiberTech.
Now, Osuji's latest study adapts the membrane for filtration in organic solutions such as ethanol and isopropyl alcohol, and its self-assembling molecules make it more efficient than traditional organic-solvent nanofiltration (OSN).
The study, published in Science Advances, describes how the uniform pores of this membrane, can be fine-tuned by changing the size or concentration of the self-assembling molecules that ultimately form the material. This tunability now opens doors for the use of this membrane technology in solving more diverse real-world organic filtration problems. Researchers in the Osuji lab, including first author and former postdoctoral researcher, Yizhou Zhang, postdoctoral researcher, Dahin Kim, and graduate student, Ruiqi Dong, as well as Xunda Feng of Donghua University, contributed to this work.
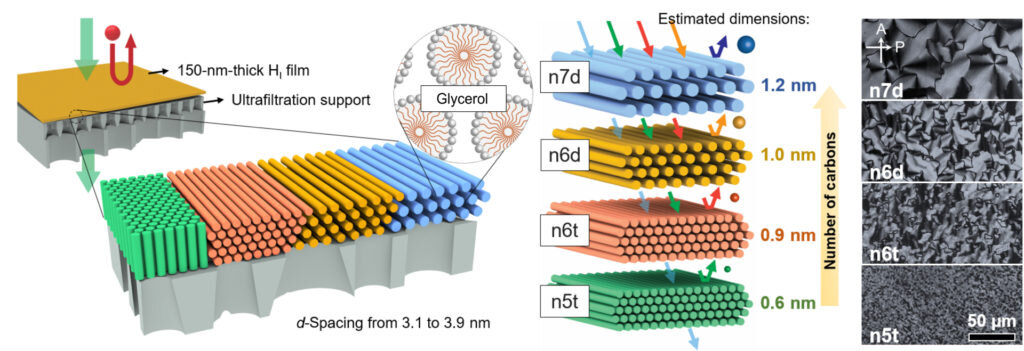
One challenge the team faced was the difficulty of maintaining membrane stability in organic solvents with different polarities. They selected molecular species, surfactants, that exhibited low solubility in organic fluids, and which could be effectively linked together chemically to provide the required stability. The surfactants self-assemble in water when they are above a certain concentration, and form a soft gel. Such self-assembly the formation of an ordered state as a function of concentration is referred to as lyotropic behavior: "lyo-" referring to the solution, and "-tropic" referring to order. The gels thus formed are called lyotropic mesophases.
The membranes developed in this study were created by forming first forming lyotropic mesophases of the surfactant in water, spreading the soft gel as a thin film, and then using a chemical reaction to link the surfactants together to form a nanoporous polymer. The size of the pores in the polymer is set by the self-assembled structure of the lyotropic mesophase.
Osuji said, at a certain concentration in an aqueous solution, the surfactant molecules aggregate and form cylindrical rods, and then those rods will self-assemble into a hexagonal structure, yielding a gel-like material. One of the ways we can manipulate the permeability, or size of the pores in our membranes, is by changing the concentration and size of the surfactant molecules used to create the membrane itself. In this study, we manipulated both of those variables to tune our pore sizes from 1.2 nanometers down to 0.6 nanometers.
These membranes are compatible with organic solvents and can be tailored to address different separation challenges. Organic solvent nanofiltration can reduce the footprint of traditional thermal separation processes. The uniform pore size of the membranes developed here provides compelling advantages in terms of membrane selectivity, and ultimately, energy efficiency as well. A specific application for this technology is in biofuel production. The isolation of water-miscible alcohols from bioreactors is a key step in the manufacturing of ethanol and butanol biofuels. Membrane separations can reduce the energy used in the separation of the product alcohols or fuels, from the aqueous medium in the reactor. The use of membranes is particularly advantageous in smaller-scale operations such as this, where distillation is not cost-effective. Additionally, the manufacturing of many pharmaceutical products often involves several steps of synthesis in different solvent environments. Those steps require the transfer of a chemical intermediate from one solvent to another miscible solvent, making this new membrane a perfect solution to drug development filtration needs.
The next steps for their research involve both theory and practice. The team plans to develop new models for membrane rejection and permeability that address the unique flow pattern of solutions through their membranes as well as identify additional future applications for their tunable technology.



0 Comments